

The chapter concludes that our understanding of metalloids toxicity will improve in the future if, in addition to the points discussed above, surface membrane potentials are considered. Approaches based on the hypothesis that metal toxicity is related to both the metal-ligand complexation processes and the metal interactions with competing cations at the cell surface (biotic ligand) have so far not been successful for assessing metalloid bioavailability. The environmental geochemical processes, factors and parameters controlling the partitioning and the speciation of B, Si, As and Sb are reviewed in relation to the bioavailability of these metalloids. The metalloid elements Ge, Te, Po and At are normally present in trace or ultratrace levels in the environment and as such are not considered of relevance in terms of environmental health. The natural abundance ofmetalloids varies from Si being the second most common element in the Earth's crust to At as the rarest of natural elements on Earth.

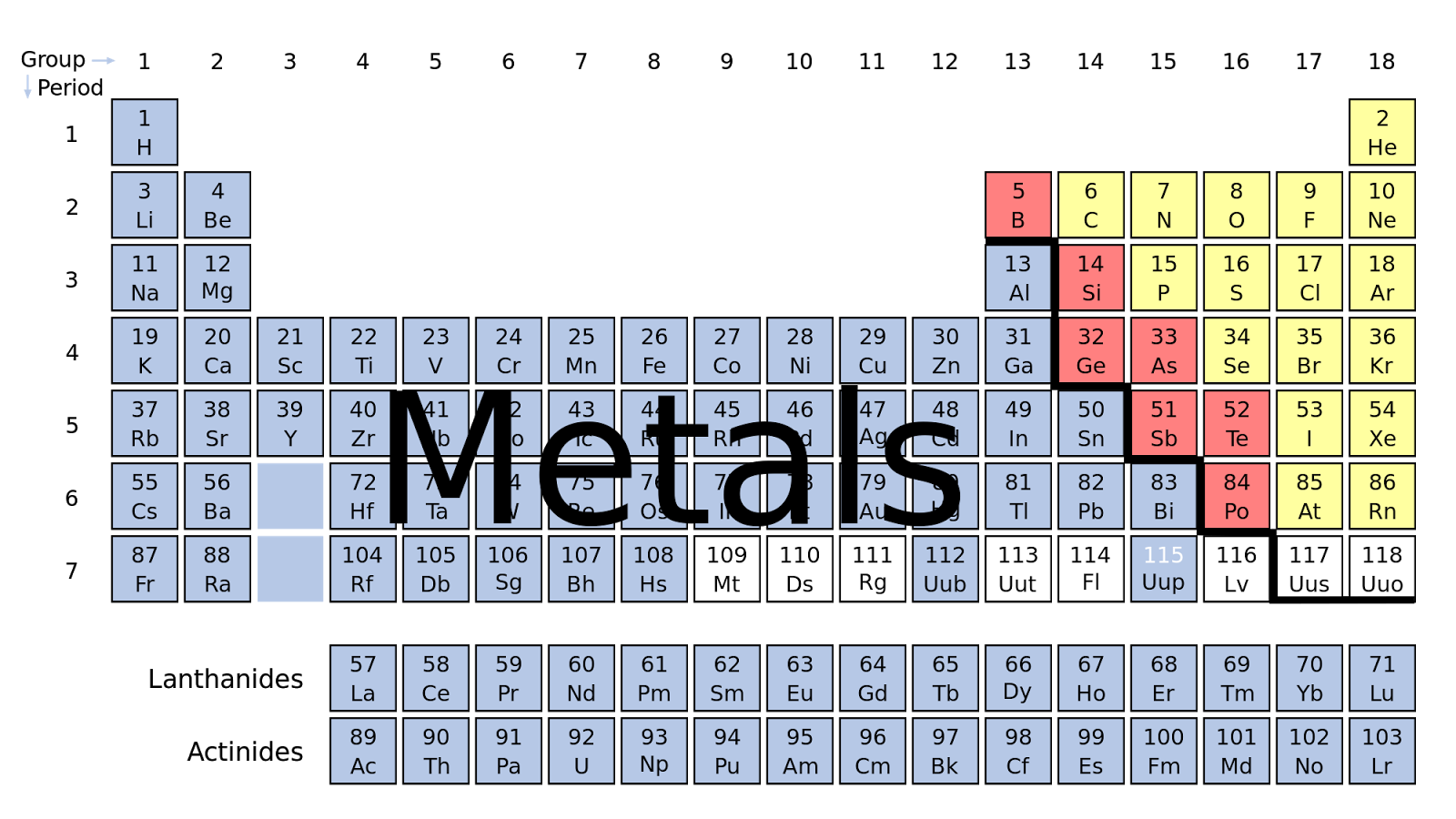
Metalloids conduct heat and electricity intermediate between nonmetals and metals and they generally form oxides. The elements boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po) and astatine (At) are considered metalloids. There are 25 metalloids in the periodic table, with the most common being bismuth, antimony, tellurium, polonium, and germanium. This chapter reviews physical chemical properties, origin and use ofmetalloids and their relevance in the environment.


 0 kommentar(er)
0 kommentar(er)
